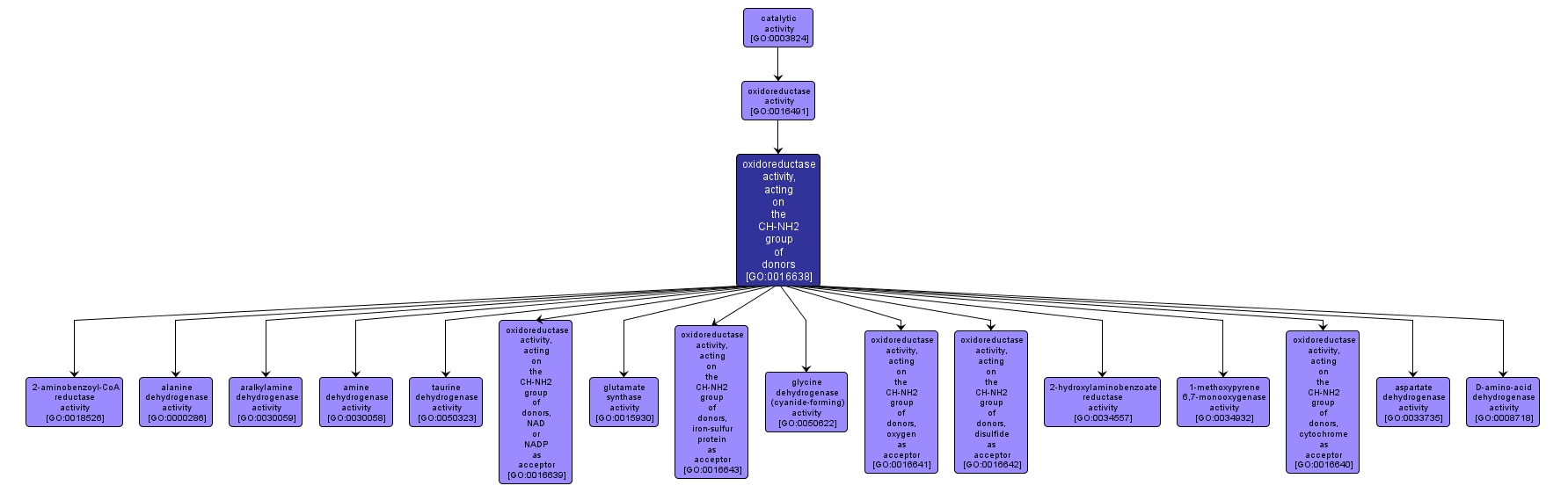
GO TERM SUMMARY
|
| Name: |
oxidoreductase activity, acting on the CH-NH2 group of donors |
| Acc: |
GO:0016638 |
| Aspect: |
Molecular Function |
| Desc: |
Catalysis of an oxidation-reduction (redox) reaction in which a CH-NH2 group acts as a hydrogen or electron donor and reduces a hydrogen or electron acceptor. |
Synonyms:
- oxidoreductase activity, acting on the CH-NH2 group of donors, other acceptors
|
|

|
INTERACTIVE GO GRAPH
|